|
Simple Daylily Hybridizing for Us Simple Folks- Part 3 (un-edited)Originally published in the Fall 2001 'AHS Journal' as "Daylily Hybridizing for Everyone" By Tom Rood, Penn Yan, NY (All daylilies in these articles are Hemerocallis ) How does daylily fertilization take place? Let's begin by reviewing the basic parts of a daylily's reproductive apparatus. The female part of the daylily is the carpel (pistil). The parts of the carpel are the stigma which is the fuzzy part with the sticky fluid on top, the style and the ovary. The style is a long "hollow" tube leading down into the ovary. The ovary contains the ovules which are the potential seeds. The pistil, upon close examination, is actually three carpels (styles and stigmas) linked together. Sometimes the styles separate making this easier to see. Each carpel leads down into its own ovary. The three seed chambers become obvious once a seed pod is opened. The male reproductive part is the stamen and consists of the pollen grains carried by the anthers. The anthers are supported by filaments. There is usually a pair of stamens for each petal/sepal combination or six for a typical daylily bloom. When a bloom first opens, the anthers are closed sealing the pollen grains. As time progresses, the anthers begin to roll open exposing thousands of ripe yellow-orange grains. Properly collected and stored, these ripe pollen grains will be good for several years. On polytepals, there is an additional set of reproductive parts for each petal/sepal combination. A four petalled bloom might then have four carpels with four ovaries instead of the usual three and an additional pair of stamens. In double daylily blooms, petaloids may form along the stamens often embedding the anthers into the petaloids. These embedded pollen grains are still capable of causing fertilization. 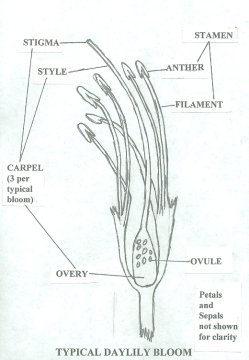 Sexual
reproduction occurs when an ovule receives hereditary
material from the pollen. This involves the union or
fertilization of the female part of the daylily and begins
when a ripe pollen grain lands upon a fertile stigma. The
process is known as pollination and can be caused by many
factors. The most important thing to remember is that each
ovule, potential seed, must be fertilized by a single pollen
grain.
Sexual
reproduction occurs when an ovule receives hereditary
material from the pollen. This involves the union or
fertilization of the female part of the daylily and begins
when a ripe pollen grain lands upon a fertile stigma. The
process is known as pollination and can be caused by many
factors. The most important thing to remember is that each
ovule, potential seed, must be fertilized by a single pollen
grain.
The sticky stigma' s fluid on top of the carpel is one of the most important keys to fertilization. It isn't always there. It may rise at another time from when the pollen ripens. In some plants this is by design to prevent self-pollination. In daylilies, the stigma's fluid usually can be observed at the same time the anthers ripen. On hot days and in extreme dry conditions, the stigma fluid may not be produced. I call this daylily reproductive stress. That is one reason why pollination success drops as the temperature rises above 85 degrees F and why many gardens in Florida erect shade houses. Irrigation is one key to helping encourage the plant to produce the stigma fluid. In the north it isn't practical to erect a shade house but if the plant is potted it can be moved into shade or some sort of a crude tent can be erected over the plant in the garden. I t is this stigma fluid that causes the pollen grain to "come alive." The fluid activates the pollen. When the pollen grain is activated, it develops a tube that begins to grow down the style towards the ovules in the ovary chamber. The tube searches out a receptive ovule, enters it and delivers two sperm nuclei. It is the pollen grain's role in life to find a single ovule and deliver these two male sperm nuclei. One sperm nuclei fertilizes the ovule which develops into the embryo or the future seed. The remaining sperm nuclei combines with two other cells to form the endosperm. The endosperm provides the nourishing tissue for the growing embryo.I have not been able to determine exactly how many ovules there are in a blossom but I think it must be at least 42. (The most seeds we've ever collected from a single diploid pod was 39). In this case, it means that 42 separate pollen grains were needed to cause fertilization to begin in 42 ovules. Now this leads to an interesting theoretical idea. If one could collect just one pollen grain from a bloom and proceed doing the same with 41 other and different blooms, each of the different 42 pollen grains could be deposited on a single fertile stigma. The result could be 42 seeds all from different pollen parents. Crazy thought isn't it? But it may help us to understand a bit more about the process. It might also explain why after we place the desired pollen on the stigmas one or more of the resulting seedlings doesn't look anything like the cross we were after. It could just be that some other pollinator added its load to ours and one or more of its pollen did its thing along with ours. As a side note, one can take the style and stigma off another bloom that has stigma fluid visible and rub it on a dry stigma. This is one way to jump start the activation of pollen grains on top of a dry stigma. After several weeks, the seed pod ripens and begins to crack open. The ripe seeds can be collected at this time. The seeds will be in three compartments mentioned above. On four petalled polytepal seed pods, there should be a fourth compartment holding additional seeds. Tetraploid daylilies are more difficult to pollinate resulting in fewer seeds than diploid daylilies. Since we know that daylily seedlings bear characteristics from both parents, we should be able to " engineer" the results. Even though hybridizing is not yet an exact science, we can both learn and have fun at the same time. One way to short circuit the route to success is to look at what has already been created and follow foot prints of hybridizers whose work we admire Those catalog pictures are pretty (naturally, to entice us to let loose with the family jewels). If time isn' t a factor, why not try making our own following the same path the hybridizer took? For us northerners that may take three or more growing seasons. We may never see the exact duplicate from our cross but we do know of at least one positive result before we even start. Every year, the AHS publishes a supplement entitled Hemerocallis Cultivar Registrations commonly referred to as the AHS "Check List." . Every five years it combines the last five years in one booklet. They are inexpensive and list the data hybridizers sent to the Registrar when registering their daylilies. Our interest is looking over the parents listed for each plant. A few hybridizers only list parents in their annual catalog so it pays to become familiar with those plant breeders of interest. Sadly, every now and then, the information gets lost in the garden and the parents become unknown. Many hybridizers are working with tetraploid daylilies. That doesn' t preclude that diploid daylilies are not worth our interest. Grace Stamile, Elizabeth Salter, and the late Ra Hansen and Pauline Henry, to name just a few hybridizers, work(ed) with diploids and with stunning results. Our only concern is to be sure we do not try to cross tetraploids with diploids. Those crosses will fail setting us back a whole hybridizing season. Putting "Tet" on the plant label for tetraploids helps keep us straight in our garden. There are two plans of attack for duplicating crosses identified in catalogs or the AHS "Check List." . The first is to simply acquire both parents and cross them together. The second is much more involved. It requires us to look at the distinctive characteristics offered by each parent, the resulting distinctive characteristics from that cross, and then going backwards in the AHS "Check List" looking at each successive parent's distinctive characteristics in all those cultivars. Unfortunately, older information is lacking for any characteristics other than the very basic bloom information. Vigour, bud counts, branching etc are all lacking in the records. Some of this is being addressed on the newest registration forms. However, many of these plants are still being grown and information is available if we know where to ask. The daylily robin is one place to search, visiting large daylily gardens is another. When we begin to evaluate the entire plant we will notice that some distinctive characteristics appeal to us more than others. What we are looking for is to identify which cultivars carry the desired distinctive characteristics, whether they are dominate or recessive and to determine the distinctive characteristic's development and amplification in successive generations. So we have to look at plants in the genetic chain of our interest. The easiest way to handle this large amount of data is to build a genealogy chart much like the one David Kirchhoff used for MING PORCELAIN (page 68, The Daylily Journal Spring 2002). Then fill in the boxes with the distinctive characteristics we observe in each plant. In this way, we will build a data base containing those traits most desired. I once built a limited chart for Pat Stamile's GLACIER BAY ('95) (ARCTIC RUFFLES X ADMIRAL'S BRAID). It went backwards all the way to the original species and nearly covered the dining room table. As I recall GLACIER BAY is only 18 or 19 generations away from the species depending which leg we went down. The only real discouragements from hybridizing come from trying to do to many different things and from setting our aspirations too high. In the beginning select only a few easily obtained goals, work with a limited number of plants, and remain focused on your objectives. Later as experience is gained the boundaries of our imaginations can be expanded. There doesn't seem to be any limit on where we are going with daylilies and what we love today will be no doubt passe in a few years. Still, the old standbys and most award winners will always have a place in our gardens. Yours may just become some of them. Bibliography: Botany, Alexander, Burnett, Zin. Golden Press, NY, NY 1970 The Clover and the Bee, Dowden. Thomas Crowell, NY, NY 1990 From Flower to Plant, Dowden. Thomas Crowell, NY, NY 1984 The Lives of Plants, Stone. Charles Scribner's Sons, NY, NY 1983 The Private Life of Plants, Attenborough. Princeton Univ Press, Princeton, NJ 1995 Biology, The World of Life 4th Edition, Wallace. Scott, Foresman, and Co. London, England 1987 |